Il contiendra diverses renseignements sur ce produit EXPERIMENTAL.
je ne suis pas l'auteur de ces documents ce ne sont que des citations et un regroupement d'informations.
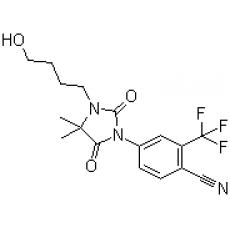
La "RU 58841" ou "PSK3841" est un nouveau topique spécifique anti-androgène, un candidat de choix donc pour le traitement de l’acné, l’alopécie androgénétique et l’hirsutisme. Il a été démontré que cette molécule appliquée par voie topique chez les animaux (étude sur les macaques) était très efficace et sans les effets secondaires des "systémiques".
La RU a été développé en France par le Dr T. Battmann et ses collègues de la Société Roussel Uclaf. Elle serait l’un des anti-androgènes le plus puissant pour un usage topique. La RU ne semble pas avoir d’absorption systémique contrairement à la finastéride et c’est la tout l’intérêt du produit car elle ne se diffuse pas dans tout le corps contrairement à la finastéride par exemple. Les effets secondaires sont donc considérablement limités. Si il y a autrefois eu beaucoup de revendications sur ce produit, ce serait le Saint-Graal des traitements topiques.
Malheureusement, pour des raisons inconnues (rentabilité?), Roussel n’a pas poursuivi la commercialisation et l’approbation de RU58841. Cela n’a pas empêché des entreprises dans d’autres parties du monde de synthétiser la même substance chimique et de la revendre à d’autres labo pour la recherche . Il faut donc commander directement aux labo, la société vous envoie le produit sous forme de poudre qu’il faut mélanger soi-même à la maison. Les droits de RU58841 ont été achetés par une société écossaise appelée ProStrakan. Le médicament est désormais officiellement dénommé PSK3841.
Avertissement: la RU 58841 est un médicament topique expérimental et n’a pas été approuvé pour utilisation chez les humains. En outre, il ya peu de données disponibles sur son innocuité. Nous n’encourageons pas ou n’approuvons pas l’utilisation de ce médicament expérimental et cet article est présenté à titre informatif seulement.
La RU est un inhibiteur des récepteurs androgènes. Elle bloque 70% des liaisons de la DHT dans le cuir chevelu. Cette molécule exerce des effets allostériques et peut fournir un mécanisme d’action unique dans le traitement de l’alopécie. Si le finastéride a été efficace pour vos cheveux, la RU le sera assurément elle aussi sans les ES (sauf à trop forte dose) certains témoignage nous venant des Etats Unis affirment que son utilisation serait plus efficace que la dutastéride (un analogue de la finastéride). Une étude sur les rats montre également que cette molécule exerce également une puissante inhibition des glandes sébacées.
le mécanisme d'action de RU.

About RU 58841
RU 58841 is a very powerful topical anti-androgen originally developed by the Roussel Corporation of France.
One of the major problems with oral anti-androgens such as Spironolactone and Flutamide is that while they are good for hair loss, they can cause all sorts of problems related to having low androgens in your body, such as loss of muscle, increased fat, loss of sex drive, gyno, etc.
RU 58841 has been shown in animal studies to be extremely powerful as a topical anti-androgen with no systematic side effects.
RU 58841 was bought by the Aventis Drug company and it was scheduled to undergo testing as a hair loss drug. Unfortunately in 1999 the scheduled trials for alopecia were discontinued and Aventis has not shown an interest in bringing the drug to market.
This turn of events has created some frustration for hair loss sufferers and there have been a few reports of people having it custom made for them. This is not a simple process and can cost in excess of $20,000 for a small batch. We were recently contacted by someone who claimed to have gained access to a supply of this drug at great expense and had decided to start using it.
We have decided to follow his progress since this is the first time it has ever been documented in humans. We do not encourage taking this drug since it has not been approved or tested in humans. The individual, who we will call "John", has undertaken this course of action of his own free will and we are documenting his experiences for scientific purposes only.
RU 58841, a new specific topical antiandrogen: a candidate of choice for the treatment of acne, androgenetic alopecia and hirsutism.
Battmann T, Bonfils A, Branche C, Humbert J, Goubet F, Teutsch G, Philibert D.
Centre de Recherches Roussel Uclaf, Romainville, France.
A new topically active non-steroidal antiandrogen, RU 58841 has been synthesized. It displays high affinity for the hamster prostate and flank organ (F.O.) androgen receptors. In vivo, when topically applied, it exerts a potent dose-dependent regression of F.O. area at a dose as low as 1 microgram/animal while being devoid of antiandrogenic activity on deep accessory sex organs and of any effect on testosterone level up to 100 micrograms/animal. In the same species, after subcutaneous administration, it induces at the dose of 300 micrograms/animal, a small decrease in F.O. area equivalent to that of 1 microgram applied topically and a weak systemic activity. In intact rats, no effects were observed up to 1 microgram/animal whatever the route of administration. These results suggest that RU 58841 might useful for the topical treatment of androgen-dependent skin disorders such as acne, androgenetic alopecia and hirsutism.
Local inhibition of sebaceous gland growth by topically applied RU 58841.
Matias JR, Gaillard M.
Nova Biosciences, Norrie Point Research Station, Staatsburg, New York 12553, USA.
The biological activity of a series of nonsteroidal, pure androgen receptor inhibitors was compared using the Syrian hamster ear skin sebaceous gland model. RU 58841, RU 56187, RU 38882 and cyproterone acetate were applied topically for 4 weeks on the ventral ear pinna of sexually mature male Syrian hamsters. Their order of efficacy was as follows: RU 58841 > RU 56187 > RU 38882 > cyproterone acetate. Maximal reduction of 60% in the size of the sebaceous glands was observed in hamsters treated with RU 58841 at a dose of 10 micrograms per day. This degree of inhibition occurred without any systemic side effects as shown by the absence of inhibition on the contralateral untreated ear pinna. Longer treatment did not produce greater inhibition since extending the treatment period from 4 weeks to 12 weeks showed similar data. The effect of RU 58841 was reversible since the inhibited sebaceous glands returned to normal size within 4 weeks after the cessation of the topical applications. The potent localized inhibition of sebaceous glands by RU 58841 demonstrates the excellent potential of this compound as a topical drug for the treatment of acne and other androgen-mediated disorders.
Inhibition of hair growth by testosterone in the presence of dermal papilla cells from the frontal bald scalp of the postpubertal stumptailed macaque.
Obana N, Chang C, Uno H.
Regional Primate Research Center, University of Wisconsin, Madison 53715-1299, USA.
Hair-follicle regression in the bald scalps of stumptailed macaques develops after puberty, which corresponds to an elevation of serum testosterone and dihydrotestosterone. Using the cultured cells from the pre- and postpubertal macaques, we examined the role of dermal papilla cells in testosterone-induced inhibition of outer root sheath cell proliferation. Testosterone showed no effects on proliferation of either dermal papilla cells or outer root sheath cells cultured alone. Testosterone-induced inhibition of outer root sheath cell proliferation occurred only in coculture with dermal papilla cells derived from the bald scalps of adult macaques but not with dermal papilla cells from the hairy occipital scalps of adult macaques or the prebald frontal scalps of juvenile macaques. Furthermore, RU 58841, an androgen receptor blocker, antagonized this testosterone-elicited inhibition. Together our data indicate that the inhibitory effect of testosterone on proliferation of epithelial cells is age dependent, and androgen may play an essential role in hair growth either by inducing repressor(s) from dermal papilla cells, which may then inhibit the growth of epithelial cells of the hair follicle, or by inducing growth factor(s) from dermal papilla cells, which, in turn, may trigger the induction of some repressors in epithelial cells, thereby inhibiting the epithelial cell growth. Our animal studies also showed that RU 58841 has a dramatic effect on hair regrowth in the bald frontal scalp of the stumptailed macaque, which may further support our in vitro culture studies showing that antiandrogens can antagonize testosterone-elicited hair growth. In summary, our studies may provide a model for further isolation of androgen-regulated repressor(s)/growth factors, which may help control hair growth and baldness.
Importance of sebaceous glands in cutaneous penetration of an antiandrogen: target effect of liposomes.
Bernard E, Dubois JL, Wepierre J.
Laboratoire de dermophamacologie, Faculte de Pharmacie, Chatenay-Malabry, France.
The significance of the sebaceous gland pathway in the cutaneous permeation of an antiandrogen, 4-[3-(4-hydroxybutyl)-4,4-dimethyl -2,5-dioxo-1-imidazolidinyl]-2-(trifluoromethyl)benzonitrile (RU 58841), was studied with normal hairless rat skin and an induced scar hairless rat skin without sebaceous glands. RU 58841 was dissolved in an alcoholic solution and encapsulated in liposomes for comparison. After 24 h, the cumulative percentage of RU 58841 absorbed in vitro was 3-4-fold higher in the normal skin than in the scar skin; in the case of liposomes, the accumulation of the drug in the normal dermis was significantly higher than in the scar one. In the in vivo cutaneous distribution, the epidermis and dermis of the normal skin contained higher amounts of RU 58841 than the scar skin (ninefold with the solution and 16-fold with liposomes). An autoradiography study showed that with the solution, the drug was mainly localized in the stratum corneum/epidermis, and with the liposomes, the drug was mainly localized in the sebaceous glands. We concluded that the sebaceous glands constituted the main pathway for RU 58841. The alcoholic solution encouraged the localization of the drug into the stratum corneum, whereas liposomes targeted the sebaceous glands.
Effects of topical antiandrogen and 5-alpha-reductase inhibitors on sebaceous glands in male fuzzy rats.
Ye F, Imamura K, Imanishi N, Rhodes L, Uno H.
Wisconsin Regional Primate Research Center, University of Wisconsin, Madison 53715-1299, USA.
The fuzzy rat, a genetic mutant between hairless and hairy albino rats, expresses androgen-dependent hypersecretion of sebum and hyperplastic sebaceous glands. Using this model for human acne, we examined the effects of inhibitors of human steroid 5 alpha-reductase isozymes, type I (MK 386) and type II (finasteride), and an androgen receptor blocker (RU58841) on regression of glandular and ductal hyperplasia. The above three agents, 1% weight volume, were dissolved into the vehicle (propylene glycol, alcohol and water) and applied on the backs of peripubertal male rats for 2 months. Control and castrate groups received vehicle alone. At 8 weeks, we examined the size the sebaceous glandular lobules and ducts in split epidermal preparations as well as in frozen sections of skin stained with osmium-potassium dichromate solution. The number of bromodeoxyuridine (BrdU)-positive cells was counted in the glandular lobes in split-skin tissues stained with BrdU immunochemistry. The results revealed that the sizes of both lobes and ducts in castrates were 40-60% smaller than in controls. RU58841 induced glandular and ductal regression equivalent to that in castrates. Finasteride induced a moderate degree of lobular and ductal reduction, whereas MK386 caused only ductal regression. Reduction of BrdU-positive cells in the sebaceous lobes was found in the skin treated with finasteride and RU58841. Serum concentrations of testosterone and dihydrotestosterone showed no significant changes in all drug-treated rats. The weight of the prostatic lobes was reduced significantly in rats treated with finasteride but not by the other two agents. RU58841 effectively counteracted endogenous androgens resulting in a suppression of growth of the sebaceous glands but not the prostate. This rodent model for androgen-dependent hyperplasia of the sebaceous glands is useful for the study of many pharmacological aspects comprising the rate of percutaneous absorption, stability and affinity to target organs of the testing compounds, and selection of adequate vehicle for topical application.
A controlled study of the effects of RU58841, a non-steroidal antiandrogen, on human hair production by balding scalp grafts maintained on testosterone-conditioned nude mice.
De Brouwer B, Tetelin C, Leroy T, Bonfils A, Van Neste D.
Skin Study Centre, Tournai, Belgium, France.
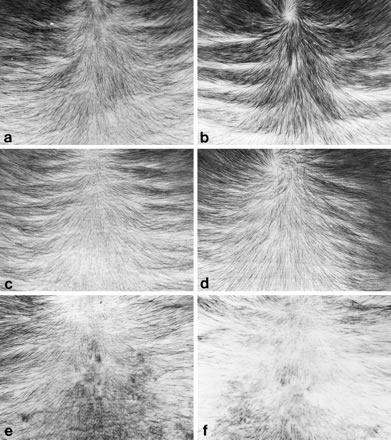
Human hair growth can be monitored for several months after the transplantation of scalp samples from men with androgen-dependent alopecia on to female nude mice. Hair production from balding sites has been shown to be inhibited in testosterone-conditioned nude mice. We used this recently reported model to study the effect of a new non-steroidal antiandrogen-RU58841-on human hair growth. Twenty productive scalp grafts from balding men were maintained for 8 months after grafting on to nude mice, and hair production was monitored monthly for 6 months. All mice were conditioned by the topical application of testosterone (testosterone propionate, 300 micrograms in 10 microL; 5 days/week) on the non-grafted flank. The scalp samples were divided equally according to the estimated hair production potential, which was based on the amount of hair present on the scalp samples before grafting. Each of the two equal groups of grafts was further allocated at random to be treated topically (5 days/week) with blinded solutions of either RU58841 1% in ethanol, or ethanol as a control. Twenty-eight active follicles appeared on the 10 control grafts. Among them only two follicles (7%) initiated a second hair cycle. However, the 10 RU58841-treated grafts bore a total of 29 active follicles, and eight of them (28%) showed a second cycle. The values for the linear hair growth rates (LHGR) were significantly (P < 0.04) higher in the RU58841-treated group. Recycling and increased LHGR indicate a positive action for RU58841 on human hair growth from balding samples grafted on to testosterone-conditioned nude mice, and encourage a clinical trial to evaluate its potential in the treatment of androgen-dependent alopecia.
Evaluation of RU58841 as an anti-androgen in prostate PC3 cells and a topical anti-alopecia agent in the bald scalp of stumptailed macaques.
Pan HJ, Wilding G, Uno H, Inui S, Goldsmith L, Messing E, Chang C.
Department of Pathology, University of Rochester Medical Center, NY 14642, USA.
The effect of androgen receptor transcriptional activation by RU58841, a nonsteroidal anti-androgen, was studied in the human prostate cancer PC3 cell line by cotransfection with wild-type androgen receptor (wt AR) and an androgen-responsive reporter (MMTV-ARE-CAT) construct. Anti-and rogens, hydroxyflutamide, and Casodex, and the antiestrogen, genistein, were studied in parallel for comparison with RU58841. The wt AR was activated only by the androgen dihydrotestosterone (DHT). Neither the anti-androgens nor antiestrogen can enhance AR transcriptional activity at 10(-11)-10(-7)M in PC3 cells. Hydroxyflutamide, RU58841, and Casodex, but not genistein, displayed competitively suppressive effects on DHT activation of wt AR. The potency of RU58841 was comparable to that of hydroxyflutamide. From this result, topical application of RU58841, which is considered to be a potential therapy for skin diseases, may induce systemic side effects. However, RU58841, on topical application, revealed a potent increase in density, thickening, and length of hair in the macaque model of androgenetic alopecia, whereas no systemic effects were detected. Together our results suggest that RU58841 may have potent antagonism to the wt AR and could be considered as a topically applied active anti-androgen for the treatment of androgen-dependent skin disorders, such as acne, androgenetic alopecia, and hirsutism.
Roles of testosterone in the growth of keratinocytes through bald frontal dermal papilla cells.
Pan HJ, Uno H, Inui S, Fulmer NO, Chang C.
Department of Pathology, University of Rochester Medical Center, NY 14642, USA.
A coculture of dermal papilla cells (DPCs) from the bald frontal scalp of stumptailed macaques with keratinocytes derived from human neonatal foreskin revealed that testosterone inhibited keratinocyte proliferation, and that the antiandrogen RU58841 abolished this response. This testosterone-induced keratinocyte growth inhibition was not observed when either type of cells was cultured alone. We also examined conditioned media from the coculture system and demonstrated the identical testosterone-induced growth inhibition on keratinocytes, and this inhibitory effect was conditioned media concentration-dependent. These results suggested that the testosterone-mediated suppression on keratinocyte proliferation might proceed through some diffusible growth mediators in conditioned media. Differential display reverse transcriptase polymerase chain reaction allowed us to isolate several genes from frontal DPCs that can be either suppressed or induced by testosterone. Supervillin, a membrane-associated, F-actin-binding protein, was identified as one of the testosterone downregulated genes in frontal DPCs. Further characterization of these testosterone-target genes may reveal the mechanism by which testosterone inhibits the growth of follicular cells in androgenetic alopecia.
resultats PSK-3841: Phase II A, antagoniste des androgènes non stéroïdiens, PSK-3841 (anciennement HMR-3841 et RU-58841) d'actualité, est à l'étude en France par Proskelia pour la valeur thérapeutique possible en AGA et l'acné. Le composé a est la phase II des essais cliniques que de Janvier 2004, et a été étudié par Roussel-Uclaf et Hoechst Marion Roussel. En comparaison avec l'anti-androgène acétate de cyprotérone composé, PSK-3841 a montré une augmentation de 20% en liaison AR. Ce composé se lie à la fois au ligand et les domaines régulateurs du récepteur d'androgène. PSK-3841 exerce des effets contraignants allostériques et peut fournir un mécanisme d'action unique dans le traitement de l'alopécie. Imamura et al. a noté que ce composé empêche régression folliculaire androgénétique chez les macaques stumptailed chauve, mais il ne favorise pas la croissance des cellules folliculaires directement. Évaluations de la photographie et de folliculogram combinée PSK-3841 5% plus minoxidil 2%, ou minoxidil à 5% par rapport aux résultats précédents de la performance de chaque composé seul ont été comparés. La repousse des cheveux plus rapide en 1 mois a été observée dans le PSK-3841 5% plus minoxidil groupe de 5%, suivi de 2 mois pour le PSK-3841 5% plus minoxidil 2% groupe. A 3 mois, la repousse des cheveux était comparable pour les deux PSK-3841 et 5% de minoxidil à 5% seul. À l'intervalle de 3 mois, les chercheurs ont noté que les deux groupes combinés ont eu la repousse des poils longs-terminale qui ont persisté pour les 6 - 12 mois. Les groupes combinés également vanté plus anagène conversion et l'élargissement folliculaire plus seuls les composés individuels. Il a été conclu que les groupes de traitement combinés ont connu le plus grand impact dès le début, mais après 1 an s'était écoulé, aucune différence n'a été observée dans les groupes de traitement combinés par rapport PSK-3841 ou le minoxidil est utilisé seul. Pan et al. comparé les effets antiandrogènes de RU-58841 avec d'autres antiantrogens connus, hydroxyflutamide, Casodex ™ (bicalutamide) et Genisten dans les cellules de la prostate humaine. PSK-3841 a démontré la suppression de l'activation de DHT au récepteur des androgènes d'un concours, et d'une manière dépendante de la dose similaire aux autres anti-androgènes. Pan et al. appliquée actualité RU-58851 5% sur le cuir chevelu chauve de macaques stumptailed. Les chercheurs ont évalué les résultats de la folliculogram et ont noté une augmentation du nombre de follicules en phase anagène et une diminution des follicules télogène par rapport à la ligne de base. Les-3841-PSK groupe traité affiché progression de télogène à anagène et une augmentation de la taille du follicule par opposition au groupe de contrôle. Pas d'effets secondaires systémiques ont été signalés. PSK-3841, dosé à 100 fois l'intervalle testé thérapeutique, n'a montré aucun signe d'effets anti-androgéniques systémiques. Pan et al. décrit les résultats de RU-58841 comme induisant « des effets remarquables sur les cheveux et la repousse folliclar dans le frontal cuir chevelu chauve de macaques." Brouwer et al. étudié les effets de PSK-3841 (1%) dans les greffes de cuir chevelu humain sur des souris nude testostérone conditionné. Les greffes PSK-3841-traités ont été comparés avec des greffons controltreated. Les greffes de PSK-3841 ont montré l'augmentation du recyclage. Un second cycle de cheveux a été observée chez 28% des follicules actives des greffes PSK-3841 contre 7% dans le groupe traité par le placebo. Souris PSK-3841 traités avaient aussi des taux plus élevés de croissance des cheveux linéaires (LHGR). Cependant, PSK-3841 n'a pas d'effet sur le diamètre des cheveux. Battmann et al. étudié PSK-3841 dans le modèle de hamster intact dans lequel la zone de l'organe de flanc est évaluée; cet organe se compose principalement de tissu sébacée androgéno-dépendante. PSK-3841 1 - 100 g / animal a conduit à la fois une et la réduction de dose en fonction du temps dans la zone des organes de flanc. Aucun effet sur les niveaux T ont été observées à cette gamme de dose ou mode de livraison. En revanche, le tissu sébacée androgéno-dépendante de la zone de l'organe de flanc des hamsters controltreated augmentée. Lorsque PSK-3841 a été administré par voie sous-cutanée, les plus fortes doses de 300 et 1000? G / animaux ont été nécessaires pour obtenir les effets anti-androgènes. Battmann et al. également testé PSK-3841 par voie percutanée, sous-cutanée et par voie orale de l'administration dans le modèle de rat. Résultats signalé que les effets anti-androgéniques systémiques ont été observés qu'à la dose la plus élevée titrée de 10 mg. Les résultats des expériences décrites précédemment ont conduit les chercheurs à conclure que PSK-3841 est un « anti-androgène locale puissant » avec une marge de sécurité favorable.
voici une autre batterie de liens contenant tout les informations sur le RU.
(la plupart des réponses a vos questions se trouvent dans ces liens).
autres descriptions :
http://www.gll-getalife.com/ru58841/
http://fr.scribd.com/doc/78969133/RU-58 ... c-Alopecia
http://www.ncbi.nlm.nih.gov/pubmed/8136306
http://daedamo.com/new/bbs/board.php?bo ... %DB&page=2
http://www.hairlosshelp.com/html/RU58841experiment.cfm
resultats clinique:
http://www.controlled-trials.com/ISRCTN71083772/
http://www.urmc.rochester.edu/george-wh ... /cv110.pdf
expérience d'utilisateurs et diverses échanges sur les forums.
http://www.hairlosshelp.com/forums/mess ... adid=89505
http://www.international-hairlossforum. ... -t210.html
http://www.hairlosshelp.com/forums/mess ... ight_key=y
http://www.hairlosshelp.com/forums/mess ... TMP=Linear










 .
.


